DBPs in Swimming Pools
UV irradiation
Formation of disinfection by-products (DBPs) in swimming pools is a known health concem. Due to its efficiency against pathogens chlorine is extensively used. However, it reacts with organic matter, enhancing DBP formation, which is suspected to be genotoxic. To decrease combined chlorine concentration in pools, MP UV lamps are used but these may produce DBPs. This study results in the thorough understanding of reactions and mechanisms occurring when water is exposed to MP UV light.
The aim of this study was to investigate the effect of radical initiators via addition prior to UV imadiation. The DBPs and more specific total trihalomethane (TTHM) level measured after UV and post-UV chlorination was compared to sample without UV exposure.
Results suggest that chloroforms are not formed in the photo-reactor, but afterwards when chlorine is added. Furthermore, it was found that UV irradiation accelerates DBP formation, but does not clearly increase the total amount formed. Last but not least, UV enhances the amount of bromine and therefore the formation of brominated DBPs.
Paper: Secondary formation of disinfection by-products by UV treatment of swimming pool water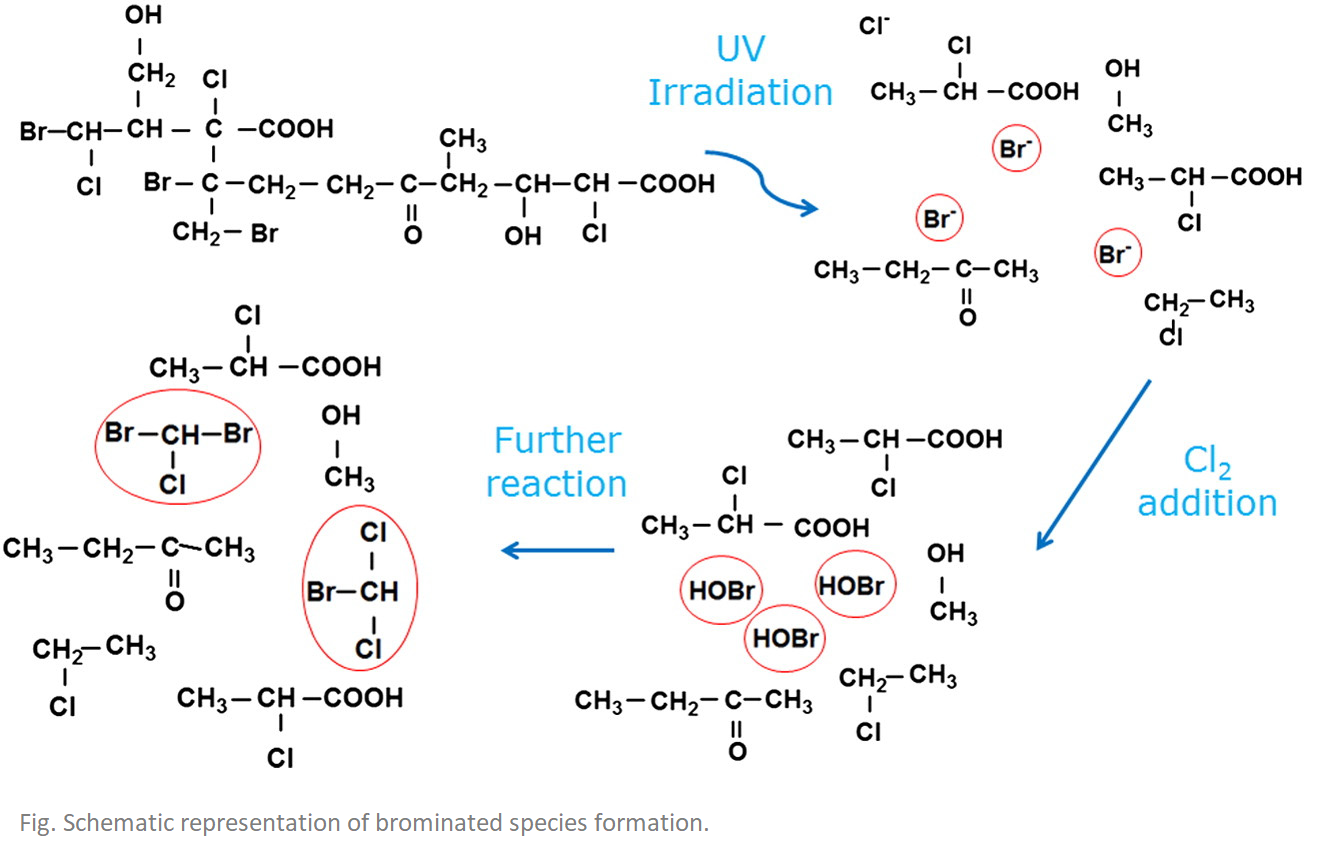

Ozonation
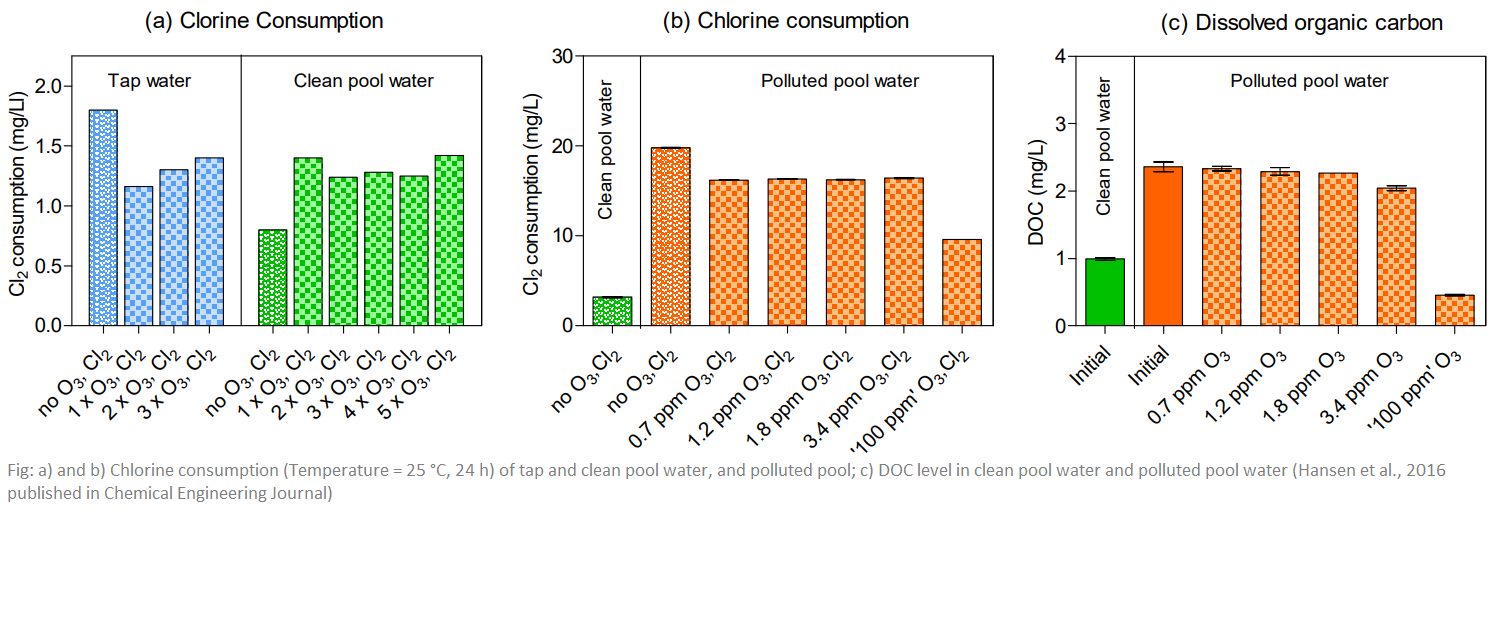
Ozonation experiments were performed using unchlorinated tap water used for filling municipal swimming pools, actual pool water and pool water polluted by addition of fresh tap water and artificial body fluid to evaluate ozone kinetics and water quality effects on formation of volatile disinfection byproducts during subsequent chlorination.
The ozone reaction was observed to behave according to first order kinetics. For tap water half-life was 4 min whilst polluted and unpolluted pool water exhibited half-life of 8 and 11 min, respectively.When ozonation dosage was repeated halflife of ozone was approximated 17-19 min in all samples.
Subsequent chlorination revealed ozone removed reactivity of dissolved organic carbon toward chlorine for tap and polluted pool water, decreasing formation rate of trihalomethanes (THM). In pool water higher rates of TTHM formation was observed after the initial ozone dosage, however this decreased with subsequent treatments. For tap and polluted pool water, ozone reacted directly with the pollutants resulting in a short ozone half-life, removing reactivity towards chlorine oxidation and preventing TTHM production. Conversely for pool water samples, due to the long half-ife of ozone, the molecule decomposed to hydroxyl radicals. These in tum reacted with aqueous organic
matter increasing chlorine reactivity and rates of TTHM formation. Formation of other non-regulated volatile byproducts (e.g. dichloracetonitrile, trichlorpropanone and trichloronitromethane) was observed to increase in pool water with ozone treatment. Thus, ozonation dosage regimes should be designed such that ozone mostly oxidizes fresh pollutants before chlorine is able to react with it.